It Never Ends – Verse Two


As I wrote in the May 10 issue of the Reader, John LaForge’s passion for fear-mongering against nuclear power (which is the safest most environment-friendly way to produce electricity), NEVER ENDS, perhaps because there’s money to be made by ignoring the science that would temper his ominous headline: “Fukushima’s “Hot Particles” Traveled Extreme Distances.” With articles like this, Mr. LaForge creates fear and paranoia, which are actually the two most common forms of radiation sickness.
Fortunately, real science about radiation is now available because everyone, including Mr. LaForge, can receive a FREE pdf of Unintended Consequences: the Lie That Killed Millions and Accelerated Climate Change by emailing tundracub@mediacombb.net, by visiting www.tundracub.com or by downloading it at http://www.unintended-consequences.org.
Those pages reveal that we are constantly bathed in radiation for our entire lives – about 2/3 from cosmic radiation and radon, with the rest contributed by consumer products like smoke detectors and by the Potassium 40 (K40) in foods like bananas and potatoes. In fact, we all radiate some 4,400 beta/gamma decays per second throughout our lives.
Mr. LaForge never admits, or doesn’t know, that just one banana produces about 15 decays per second from its potassium-40, or that our smoke detectors emit 30,000 decays per second, so when nuclear power critics fuss about 64,000 decays per second entering the ocean at Fukushima, we need to know that 64,000 emissions per second is equal to just 14 seconds of the potassium radiation activity that occurs inside our bodies every day. In addition, just one cubic meter of normal seawater radiates some 12,000 decays per second. Measured against normal background radiation, the “hot particles” that Mr. LaForge writes about will affect us about as much as adding a grain of sand to a 10-mile beach will affect its rate of erosion.

The fear of radiation that so many profit from began in the 1930s when Robert Muller caused mutations in fruit flies by exposing them to x-ray doses that were equivalent to 1,000 mammograms in just 3 1/2 minutes, which is a whopping big dose.
Muller, however, called it a small dose and claimed, against contrary evidence from his peers, that radiation was harmful all the way down to zero – an assertion he repeated when he received a Nobel Prize in the 50s when he said there could be “no doubt” about his conclusion. Muller’s lie subsequently became scientific dogma because, back then, we knew very little about the effects of radiation on DNA – the complex, spiral, chain-like molecules that contain our genetic codes.
When our radiation safety standards were set In the 50s, we didn’t know that researchers at the Massachusetts Institute of Technology would eventually discover that DNA strands can break and repair about 10,000 times per day per cell, (That is not a typo!), and that a 30-fold increase in background radiation only increases the number of breaks from 10,000 to 10,012 per day, which our repair mechanisms can easily handle.


We didn’t know that the 93,000 Japanese atom bomb survivors of Hiroshima and Nagasaki who experienced a similar 30-fold increase in radiation would have no increase in cancers over the following 50 years, and we had no way to know that those who received less than a 30-fold increase would even outlive their unexposed peers.
We didn’t know that citizens in the Indian district of Kerala were living healthy, normal lives despite constant exposures to background radiation 100 times higher than average U.S. levels.
We didn’t know, back then, that most DNA breaks are caused by ionized oxygen atoms from the normal metabolism within our cells, which is why we are told to favor anti-oxidants like grapes and greens. And we didn’t know that, because DNA is a double helix, the duplicate information in the other strand lets enzymes easily repair single strand breaks.
In fact, our cells have been repairing DNA breaks since FOREVER, and they have become very good at it. http://youtu.be/UzXcq2h0VCk?t=7m10s.
If people understood that “…we have billions of cells that die every day and must be replaced, they will be better able to accept the fact that our bodies have efficient repair mechanisms that can handle low level radiation”. SCIENCE magazine, March, 2015. (Adults have about 37 trillion cells.)
Bottom line; Muller’s lie became scientific dogma in the 50s, and dogmas are very hard to change, especially when you have individuals and industries that profit from keeping those dogmas alive.
Dr. Alex Cannara explains it this way: “Radiation from unstable isotopes is always decreasing. That’s what the “half-life” of an element expresses. Going back in time is going back to higher radiation environments — 8 times more for Uranium-235 when photosynthesis began to make oxygen common, and oxidation made elements like Uranium soluble in water. Living things, back then, were even more intimately in contact with radioactive isotopes.
“So how did life survive higher radiation, and how did it survive the increasing oxygen atmosphere, which corrodes life’s hydrocarbons into CO2 and water?
“The answer is simple: Nature evolved repair mechanisms. Each cell repairs proteins or digests badly malformed cells. Each cell repairs genetic material before it’s copied for reproduction.
“A DNA or protein molecule doesn’t know if a bond has been broken by an oxidizing radical, an alpha particle, or a microbial secretion. Our cellular-repair systems have evolved to fix defects regardless of cause. Thus, Nature has, for billions of years, been able to deal with chemical and radiation threats. However, today, chemical threats have increased because of industry, but radiation threats have actually decreased.
“Therefore, we should not be surprised by the absence of radiation deaths at Fukushima and the small death rates in and around Chernobyl.”
Regardless of Mr. Laforge’s “hot particles”, we are constantly bathed in many types of radiation, some of which are neutrinos, tiny bits of energy created by the universe – and by a particle accelerator at Chicago’s Fermi lab. There, the accelerator’s mighty arm hurls protons at a carbon target to create neutrinos, some of which raced toward a played-out iron mine beneath Soudan.
Undeterred by solid rock, the tiny neutrinos zip beneath Lake Superior so quickly that they reached Soudan just 2.5 milli-seconds after their birth, only to be captured in a huge “catcher’s mitt” in an attempt to solve two problems: Where is the missing mass of the universe that physicists believe exists, and will a better understanding of particle physics lead to a new, cheap and almost unlimited source of energy?

Novelist John Updike understood neutrinos, and he summed them up this way:
Neutrinos, they are very small,
They have no charge; they have
no mass
And do not interact at all.
The earth is just a silly ball.
To them, through which they
simply pass,
Like dust mates down a drafty hall
Or photos through a sheet of glass.
They snub the most exquisite gas,
Ignore the most substantial wall,
Cold-shoulder steel and
sounding brass,
Insult the stallion in his stall,
And, scorning barriers of class,
Infiltrate you and me! Like tall
And painless guillotines they fall
Down through our heads into
the grass.
At night they enter at Nepal
And pierce the lover and his lass
From Underneath the Bed -
you call
It wonderful; I call it crass.
Given this fascinating information, one wonders when Mr. LaForge will warns us about these omnipresent, invasive neutrinos.
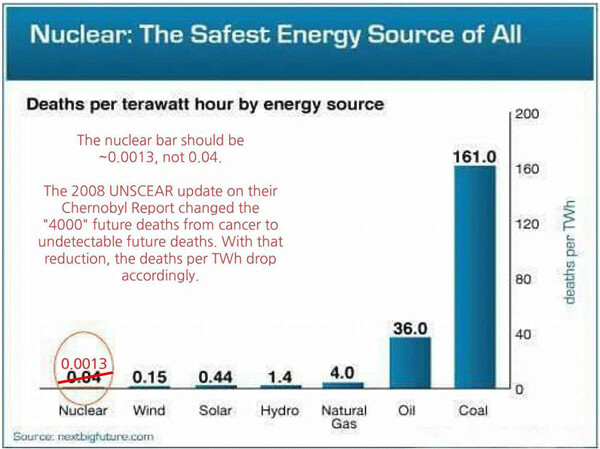
And now, like Peter Falk’s Colombo, who always had one more question, I’d like to ask Mr. LaForge to examine the two safety/health charts that accompany this column and then explain the logic of opposing super-safe, CO2-free, 24/7 nuclear power and apparently supporting deadly, intermittent, carbon-dependent, disease and disability-causing alternatives even though we now have reactors that cannot melt down and can even consume our stored nuclear “waste” as fuel.
George Erickson is a member of the National Center for Science Education and the Thorium Energy Alliance. His website is www.tundracub.com. For a presentation on nuclear power and energy alternatives, email tundracub@mediacombb.net
